|
The more sophisticated hybridization model recognized that these orbitals will be modified by their interaction with other atoms. This is a big departure from the simple Lewis and VSEPR models that were based on the one-center orbitals of individual atoms. In its full development, molecular orbital theory involves a lot of complicated mathematics, but the fundamental ideas behind it are quite easily understood, and this is all we will try to accomplish in this lesson. 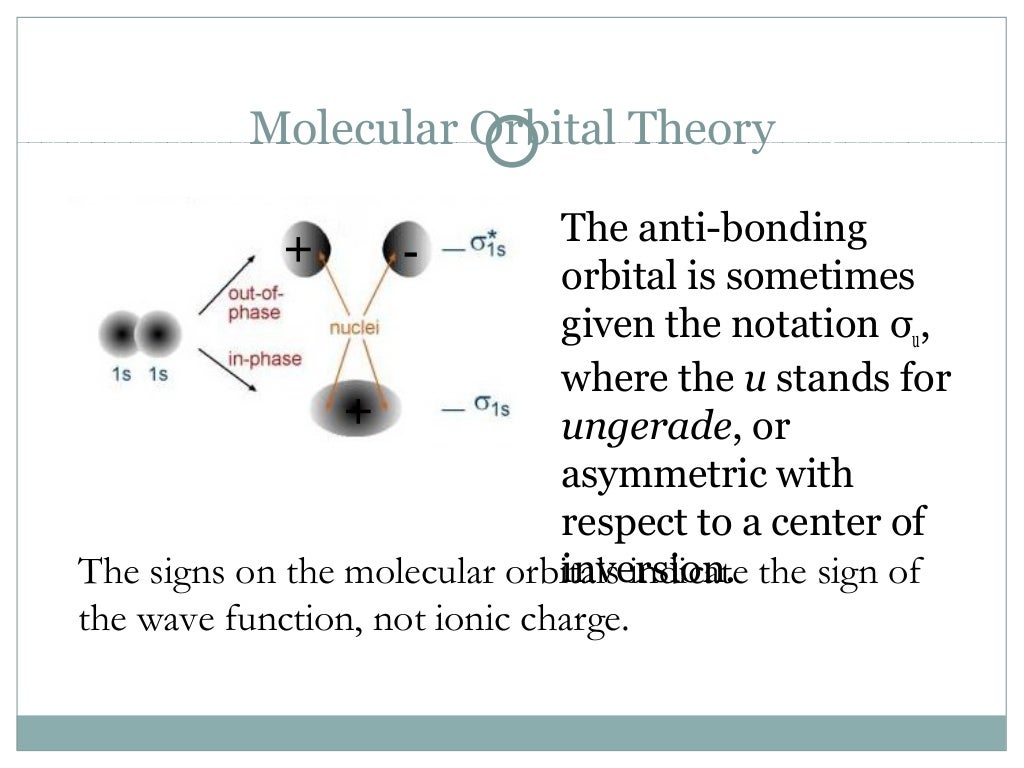
The molecular orbital model is by far the most productive of the various models of chemical bonding, and serves as the basis for most quantiative calculations, including those that lead to many of the computer-generated images that you have seen elsewhere in these units.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
